

Public misperception and excessive fear concerning nuclear radiation from nuclear energy production and waste, along with irrational government policy has led to the near demise of a very promising source of energy for the United States. These misperceptions, excessive fears, and government policies need to be reconsidered. In a logical progression the following topics will be discussed: Mass and Energy; What is Nuclear Radiation?; Nuclear Energy Production; Public Perceptions of Nuclear Radiation and Energy Production; The Benefits of Nuclear Energy; Radiation and Nuclear Energy Resources. Let's begin . . .
I. Mass and Energy
Everything in the universe can be classifeid as either Matter or Energy.
Matter: is anything that has mass and takes up space. Matter has four states: solid, liquid, gas, or plasma.
If it isn't matter then it is . . . energy.
Energy: it is often easier to say what energy can do than to say what it is. Energy can do work. Work in physics is defined as, Work equals force times distance (W = F x d), and is measured in SI units called joules (J). Energy can make changes in matter, or itself (such as transforming from one form of energy into another form of energy).
There are many forms of energy: Mass Energy, Electromagnetic Spectrum Energy (Radio waves to Gamma waves), Chemical Energy, Heat Energy, Potential Energy, Kinetic Energy . . .
The Form of Energy we are interested in is Mass Energy.
As discovered by Albert Einstein, matter and energy have a very special relationship. In fact any matter (that by definition has mass) has a tremendous amount of energy locked-up inside.
This amount of energy can be calculated with Einstein's famous equation:
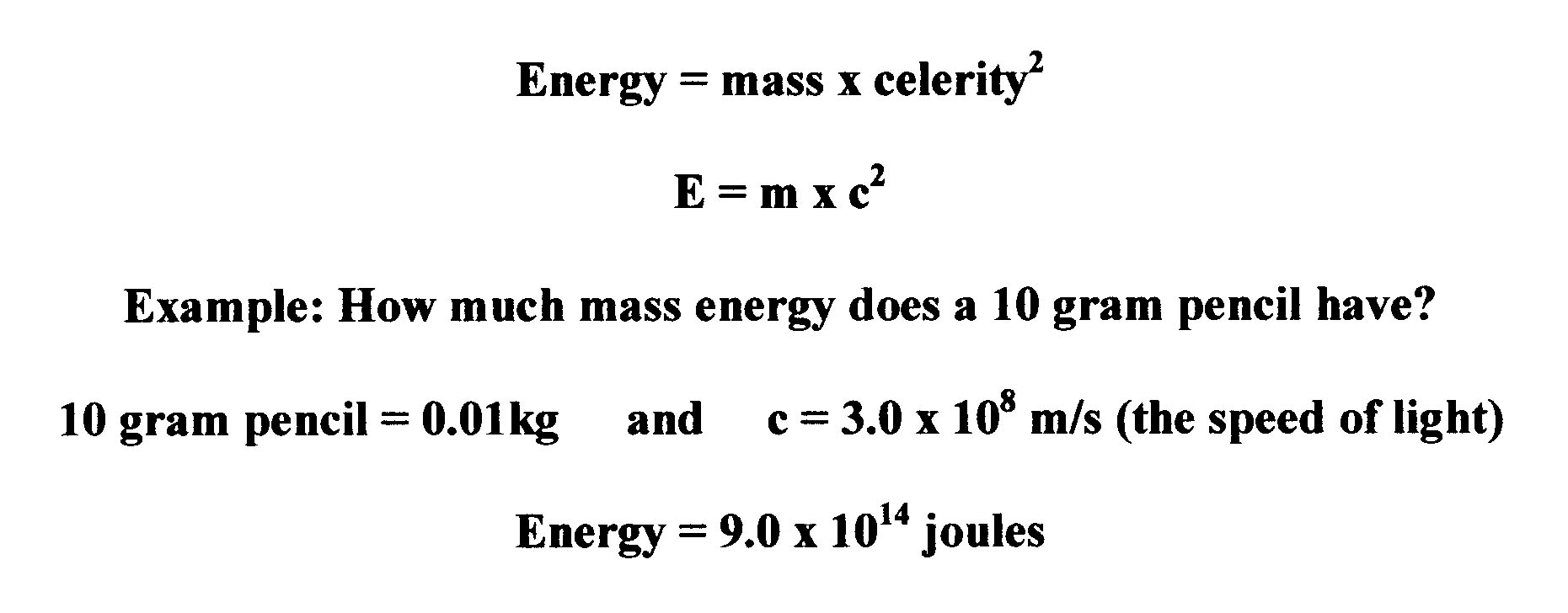
Turns out that under ordinary circumstances we cannot get to this energy. As Einstein said, "It is like a fabulously rich man that saves and never spends any money." Most forms of matter are stable. However, there are some forms of matter that are naturally unstable and spontaneously decay releasing this energy in the form of kinetic radioactive sub-atomic particles. Elements 92 and above (known as transuranic elements) on the Periodic Table, as well as some isotopic forms of elements below 92 are naturally radioactive.
Isotope: A form of an element having a particular number of nuetrons in the nuclei of its atoms. Different isotopes of a particular element have the same atomic number but different atomic mass numbers.
II. What is Nuclear Radiation?
Radiation: (a) Energy transmitted by electromagnetic waves (radio to gamma waves; v = f x l ) (b) The particle given off by radioactive atoms such as uranium.
We are interested in definition (b).
Radioactive: Term applied to an atom with a nucleus that is unstable and that can spontaneously emit a particle and become the nucleus of another element.
The principle kinds of radioactive particles that are emitted from unstable atoms are: (Source: Conceptual Physics, Paul G. Hewitt)
Nuetron: Nuetrons have mass and as the name suggests are nuetral in charge. Nuetrons can decay out of an unstable nuclei with considerable kinetic energy, and be reabsorbed by other nuclei resulting in nuclear reactions. Nuetrons are difficult to stop and require lead or other heavy shielding to block them. A lone nuetron will spontaneously decay into a proton plus an electron (and also an antineutrino, a tiny particle we will not discuss here). Out of a bunch of lone nuetrons, about half of them will decay in 11 minutes. Particles that decay in this or similiar ways are said to be radioactive. A lone nuetron is radioactive. By the mass-energy equivalence, the mass of a nuetron is slightly greater than the total mass of a proton plus electron. So when a nuetron decays into a proton and electron, there is less mass after decay than before. The difference is energy.
a: Alpha particle. Alpha particles have a positive electric charge and are made of two protons and two neutrons and are identical to the nuclei of helium atoms. Helium is a by product of alpha decay. Alpha decay will change an element (transmutation) by decreasing the atomic number by two and the mass number by four (since mass number is # of protons + # of nuetrons). Alpha partilcles are the easiest to stop. They can be stopped by a reasonably heavy piece of paper or a few sheets of thin paper.
b: Beta particle. An electron is ejected from the nucleus when a nuetron is transformed into a proton. Beta decay will change an element (transmutation) by decreasing the mass number by one, and increasing the atomic number by one and ejecting an electron. Beta particles have a negative charge. Beta particles go right through paper but are stopped by several sheets of aluminum foil.
g: Gamma photon. A gamma ray (many gamma photons streaming) is massless energy. Like visible light, gamma rays are simply photons of electromagnetic radiation, but of much higher frequency and carry a large amount of energy. Gamma rays are emitted when nucleons jump from one orbit to another of lower energy (similiar to how electrons form visible light). See Electromagnetic Spectrum. Gamma rays are difficult to stop and require lead or other heavy shielding to block them.
Cosmic rays: The earth's atmosphere is continuously bombarded from above by cosmic rays---mainly high-energy protons along with other atomic nuclei---from beyond the earth. The magnetic field and atmosphere of earth acts as a protective shield, but some cosmic rays penetrate to the surface. At higher altitudes cosmic radiation is more intense. (Examples: living in Denver you recieve 2x the normal amount at sea level; flying 2x across the US is equivalent to a chest X-ray).
Nuetrino: We are bombarded most by what harms us least --- nuetrinos. They are weak interacting particles, have nearly zero mass, no charge, and are produced frequently in radioactive decays. They travel at the speed of light, passing unhindered through our bodies by the billions every second. They pass completely through the earth with only occasional encounters. About once per year on the average, a nuetrino triggers a nuclear reaction in your body. For the most part they are harmless.
Now that we know a little about these radioactive sub-atomic particles, let's look at a poster produced by General Atomics that explains it in better detail and assesses the risks associated with natural and man-made sources of radiation. Keep in mind, no matter the source (from man-made or natural) all radiation is the same. There is no difference.
General Atomics Radiation, Radioactivity and Risk Assessment Poster
See also:
Lawrence Berkeley National Laboratory: The ABC's of Nuclear Science
PBS Frontline: Facts About Radiation
For animated Halflife Sequences for Pu241, U238, U235, and Th232 see:
Use Geiger Counter to measure background radiation, as well as several commonly available weak radioactive sources.
III. Nuclear Energy Production
There are two principal types of nuclear reactions --- fission and fusion. Fusion, the process by which our sun releases energy has an incredible promise for delivering vast amounts of energy in our future. But since it is still experimental we won't discuss it here, but look into it on your own. See: General Atomics Fusion Group and Virtual Tokamak.
The other type of nuclear reaction is fission. Fission is a nuclear process in which a heavy nucleus splits into two smaller nuclei. An example of a fission reaction that was used in the first atomic bomb and is still used in nuclear reactors is
The products shown in the above equation are only one set of many possible product nuclei. Fission reactions can produce any combination of lighter nuclei so long as the number of protons and neutrons in the products sum up to those in the initial fissioning nucleus. As with fusion, a great amount of energy can be released in fission because for heavy nuclei, the summed masses of the lighter product nuclei is less than the mass of the fissioning nucleus. Fission occurs because of the electrostatic repulsion created by the large number of positively charged protons contained in a heavy nucleus. Two smaller nuclei have less internal electrostatic repulsion than one larger nucleus. So, once the larger nucleus can overcome the strong nuclear force which holds it together, it can fission. Fission can be seen as a "tug-of-war" between the strong attractive nuclear force and the repulsive electrostatic force. In fission reactions, electrostatic repulsion wins.
The energy that is released by the fission
of one U235 atom is enormous---about seven million times the energy
released by the explosion of one TNT molecule. This energy is
mainly in the form of kinetic energy of the fission fragments,
with some energy given to ejected nuetrons, and the rest to gamma
radiation.
Fission is a process that has been occurring in the universe for billions of years. As mentioned above, we have not only used fission to produce energy for nuclear bombs, but we also use fission peacefully everyday to produce energy in nuclear power plants. Interestingly, although the first man-made nuclear reactor was produced only about fifty years ago, the Earth operated a natural fission reactor in a uranium deposit in West Africa about two billion years ago! (Source: LBN Laboratory)
It is also necessary to understand the concept of critical mass without which a fission reaction will not occur.
Critical mass: The minimum mass of fissionable material in a nuclear reactor or nuclear bomb that will sustain a chain reaction.
Lawrence Berkeley National Laboratory: The ABC's of Nuclear Science
PBS Frontline: Get Close to a Nuclear Fission Reactor
The Quick Virtual Nuclear Power Plant Tour
IV. Public Perceptions of Nuclear Radiation and Energy Production
(Source: PBS Frontline: "Nuclear Reactions: Why do Americans Fear Nuclear Power?" )
Public misperception and excessive fear concerning nuclear radiation from nuclear energy production and waste are real. Are these fears rational?
First take this survey to see what you think. The results may surprise you.
PBS Frontline: Nuclear Phobia Survey
How does the nuclear industry compare in safety with other industry and activities?
PBS Frontline: Disasters and Deathtolls
Some common public perceptions and beliefs:
These misperceptions and excessive fears need to be reconsidered. Please see the following interviews:
PBS Frontline: Interviews - Ralph Nader - Consumer Advocate
V. The Benefits of Nuclear Energy
The benefits of nuclear radiation and energy are many:
PBS Frontline: Interviews - Dr. Charles Till - Talks about the Integral Fast Reactor
Integral Fast Reactor (IFR) - Argonne National Laboratory-West
Argonne National Laboratory-West
Also see the following:
PBS Frontline: Why the French Like Nuclear Energy
If there are so many benefits to nuclear energy with acceptable levels of risk, why then is the U.S. nuclear industry so far behind other countries using nuclear power and at near ruin? The answer is complex, but it comes down to public perception and U.S. policy. Please see the following interviews:
PBS Frontline: Reprocessing: The End of a Technology
PBS Frontline: Interviews - Richard Stallings - Former Nuclear Waste Negotiator
PBS Frontline: Interviews - Glenn Seaborg - One of the founding Fathers of the Atomic Age
VI. Radiation and Nuclear Energy Resources
